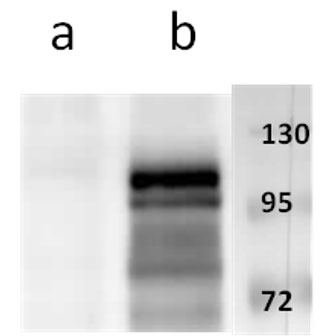
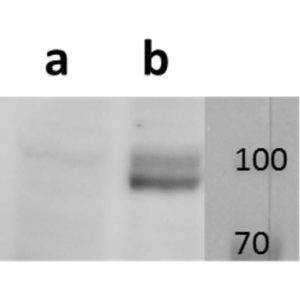
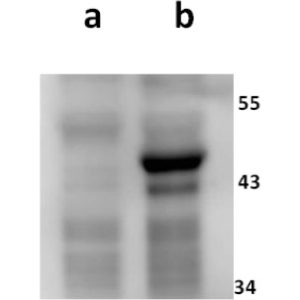
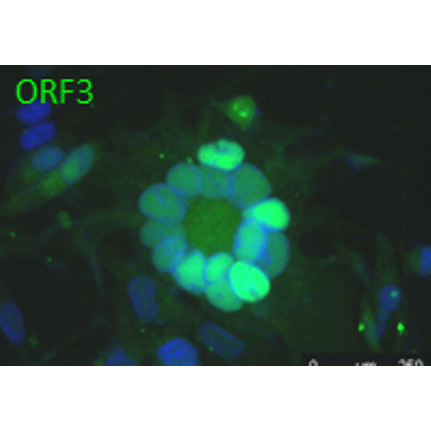
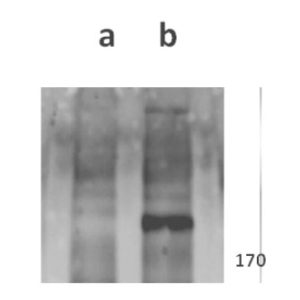
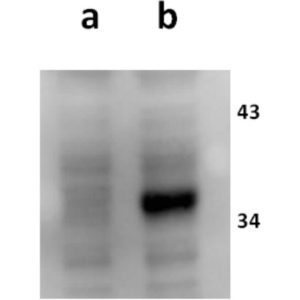
Anti-ORF11 (VZV) antibody
Clone: VZ 11N.12
Catalog No.: HR-VZV-07
Host Species: Mouse
Reactivity: Varicella zoster virus
Antigen/Immunogen: The immunogen consisted of ORF11 N terminal domain (amino acids 1-400) and was produced in E.coli
Tested Applications: ELISA, IF, WB
€200.00 – €1,500.00