Anti-UL89 (HCMV) antibody
Clone: UL89.08
Catalog No.: HR-HCMV-04
Host Species: Mouse
Reactivity: Human cytomegalovirus
Antigen/Immunogen: The immunogen consisted of UL89 N terminal domain and was produced in E.coli
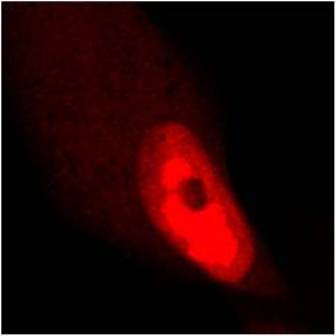
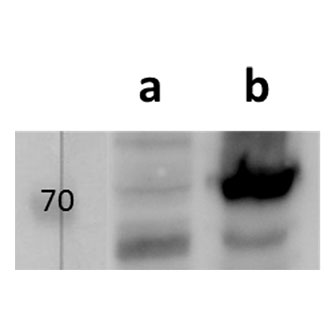
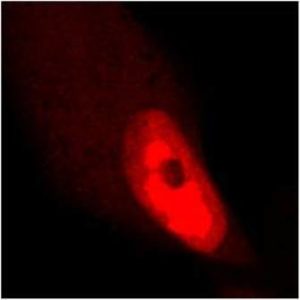
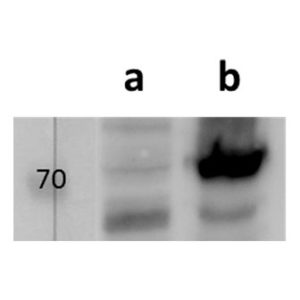
Tested Applications: ELISA, IF, WB
€200.00 – €1,500.00
Available Options:
Description
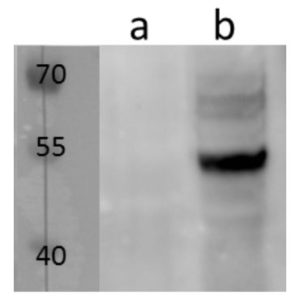
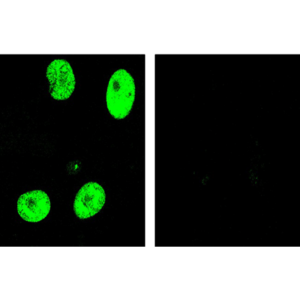
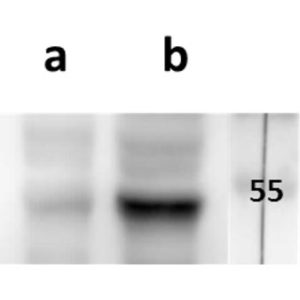
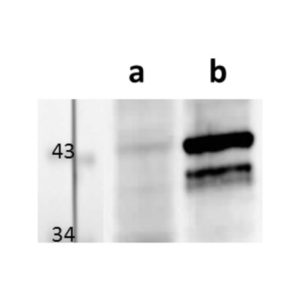
pUL89 is highly conserved 75-kDa protein. It is a small terminase subunit of Human cytomegalovirus (HCMV) able to cleave viral DNA into unit-length genomes of appropriate length for encapsidation. pUL89 is a ring-shaped protein and crystal structure analysis showed it has ability to dimerize (150-kDa) though it immunoprecipitates mainly as a 75-kDa monomer (Biochemistry (2006) 45, 8855-63). In infected cells it is translocated to the replication centers. It interacts with large 130-kDa terminase subunit pUL56 (p130) via helix α4 and this interaction enhances its nuclease activity under low protein concentrations (PNAS (2010) 107, 16078-83; Nucleic Acids Research (2002) 30, 1695-703). pUL89 has predicted ATPase activity (Antiviral Therapy (2007) 12, 217-32). It is a homolog of HSV-1 terminase subunit UL15. Our anti-UL89 antibody UL89.08 (described in J. Virol. 2013 Feb;87(3):1720-32), works in ELISA (on immunogen), in IF (on HCMV infected cells) and each LOT is validated for Western blot (of HCMV infected cell lysates), where it specifically recognizes a protein species of ~78 kDa, which corresponds well to the predicted size.
Additional information
| FORM | Liquid |
|---|---|
| STORAGE | Long term -20 °C, short term +4 °C. Avoid freeze-thaw cycles. |
| CONCENTRATION | 1 mg/mL |
| PURITY | Affinity purified |
| CLONALITY | Monoclonal |
| ISOTYPE | IgG2a |
| LIGHT CHAIN TYPE | kappa |
| REFERENCES | n/a |