Anti-ORF5 (VZV) antibody
Clone: VZ 5.12
Catalog No.: HR-VZV-01
Host Species: Mouse
Reactivity: Varicella zoster virus
Antigen/Immunogen: The immunogen consisted of full length VZV ORF5 and was produced in E.coli
Tested Applications: ELISA, WB
€200.00 – €1,500.00
Available Options:
Description
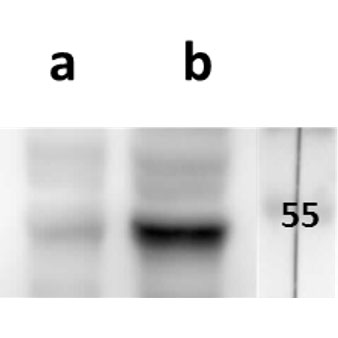
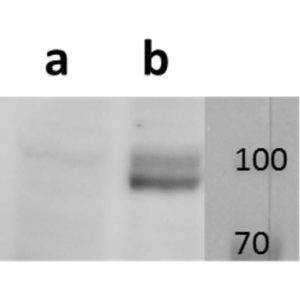
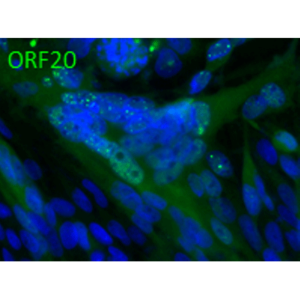
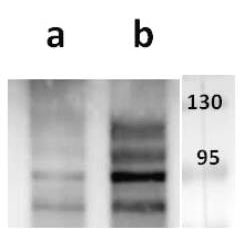
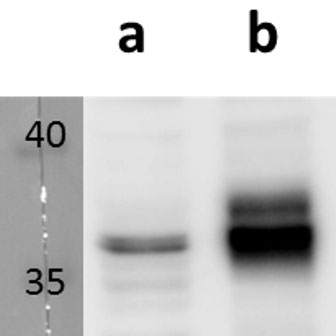
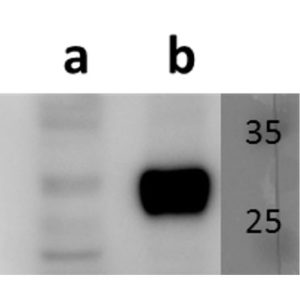
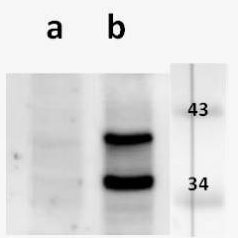
Open reading frame 5 (ORF5) of Varicella zoster encodes for envelope glycoprotein K (gK) highly conserved among alphaherpesviruses. Based on analogy to its HSV-1 homolog, VZV gK is believed to participate in membrane fusion along with other major glycoproteins. Calculated molecular mass of glycosylated form is around 40-kDa and it has two N-glycosylation sites required for the conformational integrity. VZV gK accumulates predominantly in the Golgi and it is transported to the plasma membrane of the infected cells. Afterwards, it is endocytosed from the plasma membrane and has the half-life shorter than that of other VZV glycoproteins including gB, gE and gH (Virology (2007) 358, 283-90 and Journal of Virolology (1999) 73, 4197–4207). Our anti VZV ORF5 antibody VZ5.12 works in ELISA (on immunogen), IF (on VZV infected cells) as well as is cross-reactive with SVV (Simian Varicella virus) and recognizes the glycosylated mature form of gK in both VZV and SVV. Each LOT of VZ5.12 is validated for Western blot (on VZV infected cell lysates, described in J Virol. 2013 Jun;87(12):6943-54.).
Additional information
| FORM | Liquid |
|---|---|
| STORAGE | Long term -20 °C, short term +4 °C. Avoid freeze-thaw cycles. |
| CONCENTRATION | 1 mg/mL |
| PURITY | Affinity purified |
| CLONALITY | Monoclonal |
| ISOTYPE | IgG1 |
| LIGHT CHAIN TYPE | kappa |
| REFERENCES | Lenac et al., J Virol, 2013 |